By Dr. Dragan Ledina
Department of Infectious Diseases, Split University Hospital Center, Split; Croatia
(At the recent ME Symposium organised by Bond University and the Alison Hunter Memorial Foundation in Brisbane, Australia (to which Invest in ME sponsored a UK virologist as part of our proposal for a UK Centre of Excellence) Q Fever was one of the topics of discussion. This paper from the second Journal of IiME might be worth looking at again)
(from the Journal of IiME Volume 1 Issue 1)

Summary
Background: Q fever is a common and acute but rare chronic zoonosis caused by Coxiella burnetii. Its acute form manifests as atypical pneumonia, flu-like syndrome, or hepatitis. Some authors observed symptoms of chronic fatigue in a small number of patients after the acute phase of Q fever; in many cases serological assay confirmed the activity of Coxiella burnetii infection. The effect of antibiotic therapy on post-Q-fever fatigue syndrome has not been studied in south-east Europe thus far.
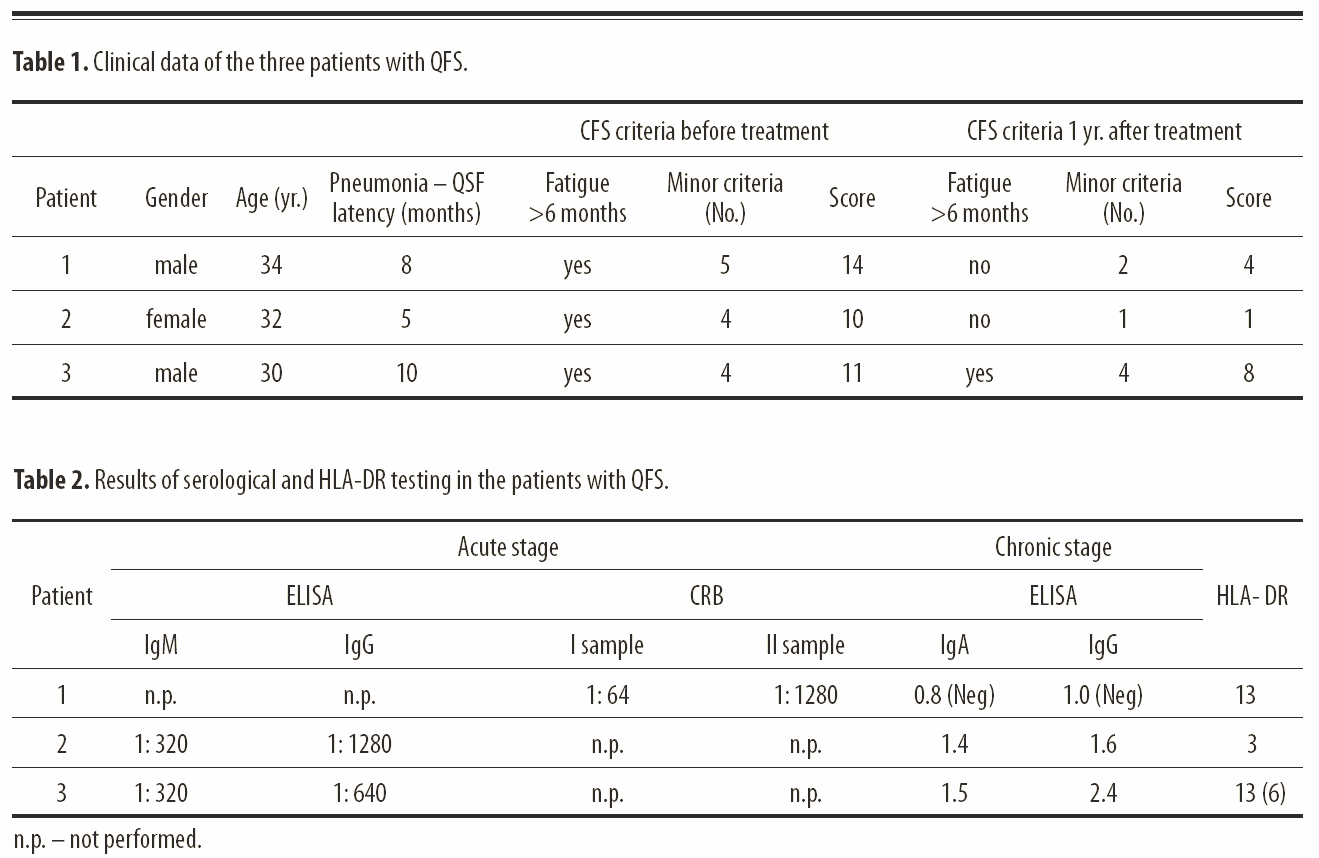
Case Reports: Three patients are presented with post-Q-fever fatigue syndrome. All fulfilled the CDC criteria for chronic fatigue syndrome. IgA antibodies to phase I of the growth cycle of Coxiella burnetii were positive in two patients and negative in one. Two patients were treated with doxycycline for two weeks in the acute phase of illness and one with a combination of erythromycin and gentamycin.
After 4–12 months they developed post-Q-fever fatigue syndrome and were treated with intracellular active antibiotics (fl uoroquinolones and tetracycline) for 3–12 months. Effi cacy of the treatment was observed in two patients, but in one patient the results were not encouraging.
Conclusions: These results suggest the possibility of the involvement of Coxiella burnetii infection in the evolution of chronic fatigue syndrome. This is the fi rst report on post-Q-fever fatigue syndrome in Mediterranean countries. Evidence of IgA antibodies to phase I of the growth cycle of Coxiella burnetii is not a prerequisite for establishing a diagnosis of CFS. The recommendation of antibiotic treatment in post-Q-fever fatigue syndrome requires further investigation.
keywords: chronic fatigue syndrome • Coxiella burnetii • post-Q fever fatigue syndrome • antibiotic treatment
BACKGROUND
Q fever is one of the most common anthropozoonoses in southeast Europe. It is caused by Coxiella burnetii, an intracellular pathogen whose classifi cation has been changed from the order of Rickettsiaceae to the order of Legionellales [1]. Human infection develops after inhalation of contaminated aerosol or consumption of unpasteurized milk. It is rarely transmitted by vectors, transfusions of contaminated blood, or transplancentally [2,3]. Recently, a major role in disease spread was attributed to air currents [4]. About 60% of infections caused by Coxiella burnetii are asymptomatic [2]. Acute infection usually presents as a febrile state, pneumonia, or hepatitis, while other organs are less commonly affected. Coxiella burnetii is endemic in rural, coastal, and non-coastal areas of southern Croatia and is associated with stockbreeding. Acute Q fever in Split-Dalmatia County (470,000 inhabitants) is most commonly presented with both pneumonia and hepatitis (60.0%), followed by pneumonia (25.8%), hepatitis (9%), and nonspecific febrile illnesses (5.2%). During the period from 1985 to 2002, 155 acute Q fever cases were hospitalized at the Split University Hospital, with a mean annual incidence of 1.82/100,000/year. All cases were verifi ed by serologic testing with C. burnetii phase II antigen as is routinely done in all patients with clinical syndrome of atypical pneumonia that live in endemic areas [5]. In the northern part of Croatia, Coxiella burnetii causes 6.45% of all interstitial pneumonias that are serologically verified [6].
In its chronic form, Q fever mostly presents as endocarditis, infl ammation of intravascular implants, osteoarthritis, and chronic hepatitis [7]. During a follow-up of convalescent patients after acute Q
We noticed that some had symptoms that were consistent with chronic fatigue syndrome (CFS). The diagnostic criteria for CFS include fatigue for six months or more together with at least four of the following symptoms: lack of concentration or/and memory that interferes with normal activities, sore throat, tender cervical or axillary lymph nodes, joint pain without swelling, muscle pain, headache, no refreshing sleep, and malaise lasting longer than 24 hours after exertion [8]. CFS is twice as common in females as in males, and it is most common between 25–45 years of age. The cause of CFS is not fully understood. There are three hypotheses about the cause of this impairment: postinfectious, immunological, and depression [9,10]. Penttila and associates found that in Australia, 20% of patients after acute Q fever develop post-Q-fever fatigue syndrome (QFS). Increased concentrations of IL-6 and interferon- as well as lowered concentrations of IL-2 that are found after stimulating peripheral blood mononuclear cells in cultures from these patients are presumed to be implicated in the pathogenesis of QFS [11].
The purpose of this paper is to emphasize the existence of CFS after Q fever in Croatia and its incidence and to show the effects of antimicrobial therapy of patients with QFS. We describe three patients who had QFS. During the period from January 2000 to December 2004, 90 patients with acute Q fever were treated at the Split University Hospital and we observed 3/90 patients with post-Q-fever fatigue syndrome. After the diagnosis of QFS was established, these patients were treated with antibiotics. They were asked to fill out questionnaires assessing their clinical condition before and after the treatment. The questionnaire survey included subjective symptoms: fatigue, lack of concentration, no refreshing sleep, sore throat, tender cervical or axillary lymph nodes, joint pain without swelling, muscle pain, headache, and malaise lasting longer than 24 hours after exertion. These symptoms were evaluated according to four grades (0: absent, 1: mild, 2: moderate, 3: severe). If the summed result of the survey was halved after the treatment, the effect of antibiotic therapy was considered favorable (Table 1).
CASE REPORTS
Case 1
A 34-year-old male shopkeeper with atypical pneumonia caused by Coxiella burnetii was treated at the Department for Pulmonary Diseases in February 2000. He did not have any serious illness before he caught Q fever. He arrived from a rural area where Q fever is endemic. Laboratory results showed an erythrocyte sedimentation rate (ESR) of 72 mm/hour, while the other hematological and biochemical parameters showed no abnormalities. The patient received a combination of erythromycin 4×500 mg/day p.o. and gentamycin 1×240 mg/day i.v for two weeks. The clinical response was good. A control chest x-ray was normal. The etiology was confirmed by the complement-binding reaction (CBR), which showed a titer for Coxiella burnetii of 1:64. A repeat CBR for Coxiella burnetii after six weeks was 1:1024.
During follow-up within the year 2000, the patient complained of disrupted sleep, morning fatigue, intense headache, prolonged fatigue lasting more than 24 hours after physical work, muscle pain, and persistent low-grade fever. Transthoracic heart ultrasound was normal. Serology for the phase I and phase II replication cycle of Coxiella burnetii did not confirm chronic infection (Table 2). After a one-year duration of symptoms, nine months of treatment with ciprofloxacin (2×500 mg/day p.o.) and doxycycline (2×100 mg/day p.o.) was instituted. The muscle pain and low-grade fever disappeared after this therapy, but the mild headache persisted. Therefore, in January 2002 a lumbar tap was performed. Cytology and biochemistry of CSF showed no abnormalities. The CSF sample was tested for Coxiella burnetii using an indirect immunofluorescence assay and the result was negative. The patient still has low intensity headache and he suffers from fatigue after physical activity, but it disappears after half an hour of rest. He has returned to work, but has changed his job from shopkeeper to watchman. He now suffers from hyperlipidemia and does not show criteria for chronic fatigue syndrome (Table 1).
Case 2
A 30-year-old male professional soldier with interstitial pneumonia was treated at the Department for Pulmonary Diseases of the Clinical Hospital of Split in February 2004.
In the acute phase of illness his ESR was 46 mm/hour, while other hematological test results were normal. Blood chemistry values were normal with the exception of AST 62 U/l (normal range: 0–29) and ALT 54 U/l (normal range: 0–30). After two weeks of treatment with doxycycline, pulmonary infiltrates resolved and hematological and other laboratory results were all within the normal ranges. IFA for Coxiella burnetii revealed positive IgM 1:64 and IgG 1:320 in a first and IgM 1:320 IgG 1:640 one month later in a second serum sample. Four months later the patient started complaining of fatigue, disrupted sleep, headaches, and muscle and joint pain. Therapy with corticosteroids was introduced and continued for one month without success. In January 2005 the patient was admitted to the Department for Infectious Diseases, and his routine hematological and biochemical tests were within physiological limits. ELISA for Epstein-Barr virus, cytomegalovirus, HIV, and Toxoplasma gondii were doxycycline for two weeks with a good clinical response, and her chest x-ray after two weeks confirmed complete regression of pulmonary infiltrations. An indirect immunofluorescence test (IFT) in the acute stage of the disease showed positive IgM (titer: 1:160) and IgG (titer: 1:640) for Coxiella burnetii. Repeated serology one month later showed IgM 1:320 and IgG 1:1280. After she had felt well for two months, she started experiencing pain in her neck. Six months later, in August 2003, in addition to the neck pain she began to suffer from insomnia, headache, sweating, and fatigue, which did not resolve after sleep. The symptoms persisted for 12 months.
She was admitted to the Department for Infectious Diseases again in October 2004. Repeated hematological and biochemical results were within physiological values. Electromyography of the upper and lower extremities showed no abnormalities and transthoracic and transesophageal heart ultrasound showed no signs of endocarditis.
Rheumatoid factor, antinuclear antibodies, and antimitochondrial antibodies as well as serology for Epstein-Barr virus, cytomegalovirus, and toxoplasmosis were negative. Anti-HIV and hepatitis B and C markers were also negative, and thyroid hormones were within normal ranges. Paired serum samples in ELISA for Coxiella burnetii showed positive phase I IgA and IgG antibodies (Table 2). The therapy included ciprofloxacin (2×500 mg/day p.o.) for two months followed by doxycycline (2×100 mg/day p.o.) for four months. The result of the six months of treatment was regression of symptoms, with only a minor headache persisting. She is now capable of doing all her housework and does not fulfill the criteria for CFS (Table 1).
Case 3
A 30-year-old male professional soldier with interstitial pneumonia was treated at the Department for Pulmonary Diseases of the Clinical Hospital of Split in February 2004.
In the acute phase of illness his ESR was 46 mm/hour, while other hematological test results were normal. Blood chemistry values were normal with the exception of AST 62 U/l (normal range: 0–29) and ALT 54 U/l (normal range: 0–30). After two weeks of treatment with doxycycline, pulmonary infiltrates resolved and hematological and other laboratory results were all within the normal ranges. IFA for Coxiella burnetii revealed positive IgM 1:64 and IgG 1:320 in a first and IgM 1:320 IgG 1:640 one month later in a second serum sample. Four months later the patient started complaining of fatigue, disrupted sleep, headaches, and muscle and joint pain. Therapy with corticosteroids was introduced and continued for one month without success. In January 2005 the patient was admitted to the Department for Infectious Diseases, and his routine hematological and biochemical tests were within physiological limits. ELISA for Epstein-Barr virus, cytomegalovirus, HIV, and Toxoplasma gondii were negative. Transthoracic and transesophageal heart ultrasound showed no signs of endocarditis. Ultrasound of abdomen was also normal. Rheumatoid factor, antinuclear antibodies, and antimitochondrial antibodies were negative. Biphasic ELISA test for Coxiella burnetii showed positive IgA antibodies in phase I (Table 2). After completing three months of antibiotic treatment with doxycycline, the patient still had fatigue, disrupted sleep, headaches, and muscle and joint pain. He still fulfills the criteria for CSF, cannot go back to work, and awaits realization of his retirement (Table 1).
DISCUSSION
Three patients with diagnoses of chronic fatigue syndrome after Q fever are described. Positive IgA antibodies for phase I of the Coxiella burnetii growth cycle suggest the possibility of chronic infection and the presence of Coxiella burnetii in macrophages [7]. Two of the patients described in this study had positive IgA antibodies for phase I of the Coxiella burnetii growth cycle and serology which was consistent with chronic Coxiella burnetii infection, while patient No. 1 had negative serology for chronic Coxiella burnetii infection (Table 2).
As there are no clinical signs or laboratory tests that could be taken as definite proof of CFS, the disease is diagnosed based on the patients’ symptoms and by excluding other diseases with similar symptoms [8]. In the last ten years, Q fever has been included in a group of diseases that are associated with the development of CFS after the acute phase of illness [7].
A recent article by Hickie et al. suggests that post-infective fatigue syndrome can occur after clinical infection by several different viral and non-viral microorganisms. The authors suggest that the CFS phenotype was stereotyped and occurred with similar incidence after Epstein-Barr virus, Q fever, and Ross River virus infection. The occurrence of CFS was predicted in the highest degree by the severity of the acute infection [12]. All our patients had moderately severe acute illness. Helbig and associates suggest a genetic predisposition for CFS[13]. Analyzing patients who had Q fever in England, Ayres and associates established that long persistence of fatigue, increased sweating, blurred vision, and shortening of breath are manifested more commonly in the group of patients that suffered from Q fever than in the control group [14]. Similar results were obtained by Marmion’s et al. [15] while comparing slaughterhouse workers who had Q fever with a serologically negative control group. Fatigue, headache, disrupted sleep, and muscle and joint pain were significantly more frequent in the group of workers with previous Q fever. Ayres [14] associated shortness of breath in patients after Q fever with possible myocardial lesions after Coxiella burnetii infection, that were first referred to by Maisch in 1986 [16]. Lovey et al. [17] established a higher incidence of cardiovascular diseases in patients who had Q fever in comparison with a control group. Later studies by Ayres et al. did not show any significant difference in cardiological measurements that would suggest cardiomyopathy or other heart diseases when comparing a group with CFS after acute Q fever and a group without symptoms of CFS [18].
Thomas et al. did not find any significant differences in the frequencies of fatigue, depression, and lack of concentration between individuals with positive antibodies for Coxiella burnetii and serologically negative individuals. The imperfection of this study was that it included all Q-feverseropositive individuals without differentiation between patients who had asymptomatic and those who had symptomatic acute Q fever, as well as the fact that the study was done on a relatively healthy population with little neuropsychiatric morbidity [19]. Although Marmion et al. suggested that the diagnosis of QFS does not require serological criteria for chronic Q fever, low serological titers against C. burnetti were associated with chronic fatigue syndrome by Penttila et al. [15,11]. It is therefore not clear if patients with symptoms of CFS and positive serology of chronic Q fever, but lacking other clinical manifestations of chronic Q-fever such as endocarditis or osteitis, as described in the cases 2 and 3 of this paper, should be included in this syndrome. We therefore believe that patients with CFS criteria, positive phase I serology, and without other clinical manifestations of chronic Q fever should be diagnosed as QFS.
Finally, is there any usefulness of antibiotic therapy of post-Q-fever CFS? The results of antibiotic therapy in patients presented in this paper were conflicting: in two cases the symptoms diminished, while the third patient continued to complain of CFS symptoms. These results are based on their clinical findings, before and after the therapy, as well as a questionnaire investigation. Up to now, there are two studies investigating the outcome of QFS therapy. Arashima et al. conducted treatment with minocycline for a period of three months in twenty patients with QFS. The result was satisfactory, and in all patients fatigue resolved, while seven patients with positive PCR test for Coxiella burnetii turned negative [20]. The limitation of this study is the absence of a placebo control group. One year later, Iwakami et al. Studied the effects of three months of antibiotic therapy in patients with post-Q-fever CFS. Although they became negative for C. burnetii DNA, in contrast to Arashima’s study no improvement of their symptoms was observed [21]. Another anecdotal attempt was the treatment of three-year-old girl with post-Q-fever CFS with interferon-g after unsuccessful antibiotic therapy [22].
The idea for such therapy was based on the knowledge that interferon-g induces the killing of monocytes infected with Coxiella burnetii. The result of treatment was satisfying and encouraging for further investigations. Although Vissar et al. [23] accentuated the diversity of the immune response of peripheral mononuclear cells in patients with CFS after stimulation with dexamethasone, our patient treated with corticosteroids did not experience amelioration of his symptoms.
CONCLUSIONS
Our case series of patients from southern Croatia, where Q fever is endemic, is in concordance with more detailed data presented in the past from other areas of the world. Therefore we can conclude that a substantial number of patients develops CFS after acute Q fever in spite of appropriate antibiotic therapy during acute infection. The results of prolonged antibiotic therapy in such the patients are inconsistent. Efforts to establish diagnostic criteria as well as therapeutic recommendations for post-Q-fever CFS require further investigation.
REFERENCES:
- 1. Brouqui P, Marrie TJ, Raoult D: Coxiella In: Murray PR, Baron EJ, Jorgenson JH, Phaler MA, Yolken RH (eds.) Manual of clinical microbiology. 8th ed. Washington D.C, ASM press; 2003; 1030–36
- 2. Marrie TJ, Raoult D: Coxiella burnetii (Q fever). In: Mandell GL, Douglas JE, Bennett JE (eds.) Principles and Practice of Infectious Diseases. 6th ed. New York: Churchill Livingstone, 2005; 2296–302 Med Sci Monit, 2007; 13(7): CS88-92 Ledina D et al – Chronic Fatigue Syndrome and Q fever CS91
- 3. Punda-Polić V, Radulović S: Sero-survey of Q fever in the north-western part of Bosnia and Herzegovina. Croat Med J, 1997; 38: 345–47
- 4. Medić A, Dželalija B, Punda Polić V et al: Q fever epidemic among employees in a factory in the suburb of Zadar, Croatia. Croat Med J, 2005; 46(2): 315–19
- 5. Lukšić B, Punda-Polić V, Ivić I et al: Clinical and epidemiological features of hospitalized acute Q fever cases from Split-Dalmatia County (Croatia), 1985–2002. Med Sci Monit, 2006; 12(3): CR126–31
- 6. Puljiz I, Kuzman I, Daković-Rode O: Clinical and epidemiological characteristics of Q fever in hospitalised patients. Infektol. Glasn, 2005; 25: 75–80
- 7. Raoult D, Marrie TJ, Mege JL: Natural history and pathophysiology of Q fever. Lancet Infect Dis, 2005; 5: 219–26
- 8. Fukuda K, Straus SE, Hickie I et al: The Chronic fatigue syndrome: A Comprehensive Approach to its Defi nition and Study. Ann Intern Med, 1994; 121: 953–59
- 9. Engelberg NC: Chronic Fatique Syndrome. In: Mandell GL, Douglas JE, Bennett JE (eds.) Principles and Practice of Infectious Diseases. 6th ed. New York: Churchill Livingstone; 2005; 120–25
- 10. Korzon M, Bukowska W, Szlogatys-Sidorkiewicz A: Chronic fatigue syndrome. Med Sci Monit, 1998; 4(2): 388–92
- 11. Penttila IA, Harris RJ, Storm P et al: Cytokine dysregulation in the post-Q-fever fatigue syndrome. Q J Med, 1998; 91: 549–60
- 12. Hickie I, Davenport T, Wakefield D et al: Post-infective and chronic fatigue syndromes precipitated by viral and non-viral pathogens: prospective cohort study. BMJ, 2006; 333(7568): 575–81
- 13. Helbig KJ, Heatley SL, Harris RJ et al: Variation in immune response genes and chronic Q fever. Concepts: Preliminary test with post-Q fever fatigue syndrome. Genes Immun, 2003; 4: 82–85
- 14. Ayres JG, Flint N, Smith EG et al: Post-infection fatigue syndrome following Q fever. Q J Med, 1998; 91: 105–23
- 15. Marmion BP, Shannon M, Maddocks I et al: Protracted debility and fatigue after acute Q fever. Lancet, 1996; 347: 977–78
- 16. Maisch B: Rickettsial perimyocarditis-a follow up study. Heart Vessels, 1986; 2: 55–59
- 17. Lovey P-Y, Morabia A, Bleed D et al: Long term vascular complication of Coxiella burnetii infection in Switzerland: cohort study. Br Med J, 1999; 319: 284–86
- 18. Ayres JG, Wildman M, Groves J et al: Long-term Follow-up of patients from the 1989 Q fever outbreak: no evidence of excess cardiac disease in those with fatigue. Q J Med, 2002; 95: 539–46
- 19. Thomas HV, Thomas DR, Salmon RL et al: Toxoplasma and Coxiella infection and psychiatric morbidity: A retrospective cohort analysis. BMC Psychiatry, 2004; 4: 326–29
- 20. Arashima Y, Kato K, Komiya T et al: Improvement of Chronic Nonspecific symptoms by long-term minocycline treatment in Japanese patients with Coxiella burnetii infection considered to have post-Q fever fatigue syndrome. Intern Med, 2004; 43: 49–54
- 21. Iwakami E, Arashima Y, Kato K et al: Treatment of chronic fatigue syndrome with antibiotics: Pilot study assessing the involvement of Coxiella burnetii infection. Intern Med, 2005; 44: 1258–63
- 22. Yutaka M, Hiroshi W, Tomoki T et al: Intractable Q fever treated with recombinant gamma interferon. Pediatr Infect Dis, 2001; 20: 547–57 23. Visser J, Blauw B, Hinlopen B et al: CD4 T lymphocytes from patients with chronic fatigue syndrome have decreased interferon-g production and increased sensitivity to dexamethasone. J Infect Dis, 1998; 177:451–54

